Custom Gene Synthesis Services
Introducing our newly enhanced Gene Synthesis
This easy-to-use solution is for researchers who want to skip in-house cloning steps and move quickly into functional studies with 100% sequence-verified clonal DNA .
Ordering
Complete synthetic genes with 100% sequence verification are delivered in a cloning or expression vector and are ready to use for a variety of applications.
- Never any reoccurring or hidden fees for custom vectors
- Transfection grade and endotoxin free options now available
- Guaranteed delivery dates
Genes in tubes
- No order minimum for tubes
- Delivered dry, normalized to 4 µg with scale up options available*
- Glycerol stocks available
Genes in plates
- Available in PCR and Echo™ plates‡
- 3 µg delivered dry or wet normalized to 20 ng/µl*
- Orders require a minimum of 24 genes per plate§
NEW Complex and long genes, simplified.
The IDT × Ansa Biosciences partnership delivers challenging gene constructs up to 50 kb with speed and reliability you can trust. Order now to get started.
Accepted sequence length and complexity will expand as IDT continues to implement Ansa's platform capabilities. If you need early access to higher complexities and genes up to 50kb, please contact IDT customer support.
* Gene sequences with added complexity can interfere with assembly and/or sequencing performance. Such sequences may result in less synthesis yield and/or additional services charges.
† The time required to manufacture a gene is dependent on length, complexity, and vector choice. >85% orders are ready to ship within the number of business days indicated. Sequences that are unstable and/or toxic to E. coli can substantially affect delivery times.
‡ Echo is a trademark of Labcyte Inc. A Beckman Coulter company
§ Plates with less than 24 fragments will incur an additional fee applied at checkout
Need your Genes faster? Check out our new Rapid Genes!
Table 1. Additional Options for Genes.
*Vectors identified as low copy vectors will have a reduced yield of 50 ug. This will be determined during the custom vector onboarding process.
IDT vectors
IDT has several catalog vectors available for use in your experiments, but if you don’t see one here that fits your needs see Custom Vector Onboarding below. We have several options for cloning vectors and vectors ready for protein expression experiments, in both bacteria and mammalian cells. When selecting our standard cloning vector option, if your insert is less than 250 bases we will use the pIDTSmart vector. All other lengths will be cloned into our pUCIDT with the antibiotic selection marker of your choice. We also offer vectors that have been optimized for use with Golden Gate Assembly; they will not have the most frequently used Type IIS restriction sites. Upon receiving your gene, you can then subclone it into the vector of your choice using a variety of methods. The identity of the cloning vector used, its sequence, and insertion site will be confirmed in the documentation that accompanies your product.
Table 2. Best-fit vectors for Genes ≤5000 bp.
| Vector name | Selection marker | Application | Sequence |
|---|---|---|---|
| pUCIDT (Amp) | Ampicillin | Cloning | TXT, GB, PDF |
| pUCIDT (Amp) Golden Gate | Ampicillin | Cloning | TXT, GB, PDF |
| pUCIDT (Kan) | Kanamycin | Cloning | TXT, GB, PDF |
| pUCIDT (Kan) Golden Gate | Kanamycin | Cloning | TXT, GB, PDF |
| pIDTSmart (Amp) | Ampicillin | Cloning | TXT, GB, PDF |
| pIDTSmart (Kan) | Kanamycin | Cloning | TXT, GB, PDF |
| pEXP-IDT | Ampicillin | Mammalian Expression | TXT, GB, PDF |
| pET-IDT | Kanamycin | Bacterial Expression | TXT, GB, PDF |
| pET-IDT C His | Kanamycin | Bacterial Expression | TXT, GB, PDF |
| pET-IDT Dual His + Thrombin | Kanamycin | Bacterial Expression | TXT, GB, PDF |
| pET-IDT N His + Thrombin | Kanamycin | Bacterial Expression | TXT, GB, PDF |
Custom Vector Onboarding NEW
If your experiment requires a vector that isn’t available in our standard genes tool, no problem. You can easily onboard your own custom vector through our website without requesting a quote.
Advantages to onboarding your vector with IDT:
- Saves you time and we do the cloning for you. Once onboarded you can order cloned genes in your vector anytime
- Onboarding has a one-time cost, no recurring fees for additional inserts
- Fast turnaround times - get your vector in as little as 10 business days
- Confidentiality is a high priority to ensure your IP is protected

Request a consultation
Have questions for our experts? Want to onboard your own vector? Your time is valuable and we’ll prioritize your inquiry. Click on “Request a consultation” to provide brief information about your project, and we’ll be in touch to discuss it ASAP.
Request a consultationProduct details
Genes and MiniGenes™ are NGS-verified, circular double-stranded DNA in a plasmid. DNA sequences 25 bp to 5 kb are provided with IDT’s in-house vectors, expression vectors, or a custom vector of your choice without additional cloning fees (see Table 1) . Sequences greater than 5 kb can be reviewed for acceptance as a custom project. Plasmids are delivered ready-to use in your preferred configuration with an accompanying QC report.
Storage conditions
| Short term (<2 weeks) | 4°C |
| Long term (up to 2 years) | –20°C |
Resources
Frequently asked questions
Will IDT store my vector?
What if my vector does not meet the requirements above?
Why can’t I order complex genes in my preferred vector?
When will you update your portal to include longer lengths and higher complexities?
We’re actively expanding the range of lengths and complexities available for online ordering. To ensure a high-quality customer experience, these capabilities are being rolled out in phases.
We will notify customers as new options become available. If you’re interested in ordering higher-complexity genes or constructs up to 50 kb today, please contact us for early access.
Why am I receiving a lower yield of DNA for my complex genes?
Ansa specializes in manufacturing highly complex genes. All genes are delivered with a guaranteed minimum yield of 250 ng and typically ship at approximately 1.5 μg (non‑normalized).
Ansa DNA quantity is measured using a fluorescence‑based method (Qubit®, Thermo Fisher Scientific), which may report lower concentrations than absorbance‑based techniques (NanoDrop®, Thermo Fisher Scientific). If you have questions about yields or quantification, we’re happy to help — please contact us for more information.
How do I order an Ansa gene?
You can now order Ansa genes directly through our online ordering portal. Each gene is automatically routed to the optimal IDT or Ansa manufacturing workflow based on length and complexity, delivering the best balance of speed and cost.
Click here to place your order now.
For sequences that exceed the current online limits, please contact us to access higher‑complexity designs and gene lengths up to 50 kb.
Are the custom genes delivered as linear fragments or in a vector?
What is required to define the insertion site for a new Custom Vector?
What type of plasmid prep is required for my custom vector?
What are the requirements for my custom vector?
- An E. coli origin of replication
- Kanamycin, Ampicillin, or Chloramphenicol resistance marker
- 1 µg of circular plasmid
A minimum of 50 bases of sequence flanking the insertion site you would like us to use for the assembly of your gene inserts into the vector. Therefore, you do not need to specify restriction sites for assembly.
How long does it take to make a custom gene?
Synthesis time depends on the length of the custom gene.
| Length of custom gene | Time to ship after order confirmation (business days) |
|---|---|
| Up to 500 bp | 8 |
| 501–5000 bp | 12 |
| More than 5000 bp | Inquire* |
* Longer sequences may take additional time depending on their length and complexity. Contact us to submit sequences longer than 5000 bp for an official review and quote, including an estimated turnaround time.
What is the length of synthetic, custom genes that IDT can synthesize?
We can synthesize custom genes from 25 bp to more than 5 kb in length.
If you would like to submit a sequence for review or obtain an official quote, contact us.
What restriction sites will be available for subcloning with my gene order?
We have removed the multiple cloning site and most common restriction sites from our standard vectors.
Because of this, you should be able to add any convenient restriction sites to your sequences to facilitate subcloning. If you would like more information about our standard vectors, contact us.
Can IDT deliver genes in custom vectors, such as one supplied by a customer?
IDT can synthesize genes in customer supplied vectors. Before we can accept orders in the custom vector, we will need to complete a validation process to confirm it is compatible with our synthesis process.
If you have questions about vector validation or would like an official quote, contact us for more details. The custom vector onboarding has a minimal fee associated, however, if the vector is accepted and successfully onboarded there are no additional fees for using a custom vector.
What are optimum storage conditions for a custom gene?
IDT ships custom gene dry unless otherwise requested. While dry they should be stable up to 2 years. After resuspending in a high-quality, molecular-grade water or buffer, pH 7.5–8.0, you should store the DNA at –20°C, and if necessary, aliquot to avoid more than 2 or 3 freeze-thaw cycles.
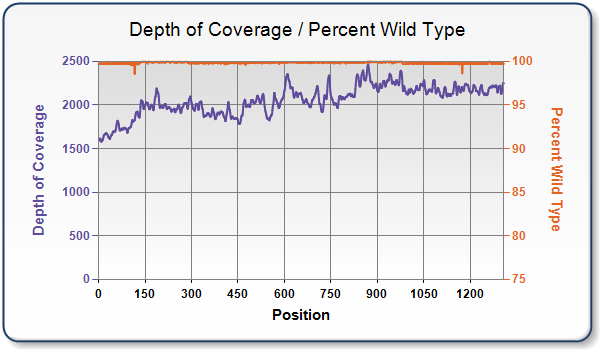
Why does the read depth vary across the sequence data for my custom gene?
Plasmid DNA is fragmented prior to sequencing. Next, the fragmented DNA must be converted to a library that is compatible with the NGS sequencing platform, which is done using transposases to randomly add sequencing adapters to the fragments.
While this process is somewhat random, it is influenced by the structure and composition of the sequence.
Similarly, the success of individual reads within the run is also influenced by structure and sequence. Highly structured areas, areas with extremes in GC, and other complexities tend to affect read depths in areas where they occur.
Why does the sequence data for my custom gene have errors?
Even with clonally pure samples, NGS traces rarely show 100% sequence identity. This is because errors do occur during sequencing reactions and the subsequent data analysis.
These error rates are typically between 0.1% and 0.01%, but they can be locally elevated in regions of extreme GC content or secondary structure. Misalignment of the traces can also occur, particularly in highly repetitive sequences.
Finally, small subpopulations of sequence variants can arise during propagation of E. coli.
What vectors are available for standard gene synthesis orders?
To optimize manufacturing and delivery time, synthetic genes ≤5 kb in length will be provided in a “best-fit” IDT cloning vector (unless the Golden Gate vectors are selected). A choice of either ampicillin or kanamycin selection markers is available. Your high-quality, synthetic gene can then be subcloned into the vector of your choice using a variety of methods.
View a list of the IDT cloning vectors and sequence information. The identity of the cloning vector used, its sequence, and insertion site will be confirmed in the documentation accompanying the product.
What is the standard delivery vector IDT uses for Genes and MiniGene™ Synthetic Genes?
To optimize manufacturing and delivery time, synthetic genes ≤5 kb in length will be provided in a "best-fit" IDT cloning vector. A choice of either ampicillin or kanamycin selection markers is available. Your high-quality, synthetic gene can then be subcloned into the vector.
For more information, and a list of the IDT cloning vectors. The identity of the cloning vector used, its sequence, and insertion site will be confirmed in the documentation accompanying the product.
Biosecurity
Sequence Information is secure and confidential at IDT. Please see our Confidentiality Statement for more information. All online ordering steps, including sequence entry and your choice of parameters, are also secure and protected.
We screen the sequence of every gene, and gene fragment order we receive to (1) identify any regulated and other potentially dangerous pathogen sequences, and (2) verify that IDT’s gene customers are legitimate scientists engaged in beneficial research.
IDT is among the five founding members of the International Gene Synthesis Consortium (IGSC) and helped to create the IGSC’s Harmonized Screening Protocol. The Harmonized Screening Protocol describes the gene sequence and customer screening practices that IGSC member companies employ to prevent the misuse of synthetic genes. IDT takes the steps set out in the Harmonized Screening Protocol to screen the sequences of ordered genes and the prospective customers who submit those orders.
For more information about the IGSC and the Harmonized Screening Protocol, please visit the website at www.genesynthesisconsortium.org.

In October 2010, the United States government issued final Screening Framework Guidance for Providers of Synthetic Double-Stranded DNA, describing how commercial providers of synthetic genes should perform gene sequence and customer screening. IDT and the other IGSC member companies supported the adoption of the Screening Framework Guidance, and IDT follows that Guidance in its application of the Harmonized Screening Protocol. For more information, please see 75 FR 62820 (Oct. 13, 2010), or https://federalregister.gov/a/2010-25728.